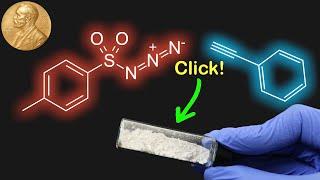
Click Chemistry in Action: The Chemistry Behind the 2022 Nobel Prize
Комментарии:

I loved the trick of washing the isolated compound laden with coloured impurities with insoluble solvents... had it been me I would have did another column chromatography again!
Ответить
Nifty and thanks. Heard about this approach. As a chemist that has dabbled in polymers, this sounds sort of familiar, as the stated objective often is to make molecules (usually up to about 5k or so monomers) of repeating monomers. Well done, altogether.
Ответить
I got to meet Dr. Sharpless today. He gave a lecture at my university talking about his work developing treatments for tuberculosis using Sulfur flouride exchange.
Ответить
How much amount (equivalent) of catalyst you have used?
Ответить
Morten Meldal is my MSc professor, great teacher and very very knowledgeable in synthesis. He came up with the "hand rule" that takes advantage of the fact that our hands are chiral, let me tell you it's sooo useful! You can go from a very complex conformation and spatial disposition of the reagents and rewrite the structure while keeping all the chiral centers correct. It saves so much time and mental energy. The hand rule alone could be enough for a nobel prize 😂
Ответить
Could you show a non enzematic way to make 1 andostenediol or 2 andostendiol? Im really looming for the process of changing its locality🎉
Ответить
👍
Ответить
Man thats why i hate column chromatography
Ответить
Barry Sharpless has already won the Nobel Prize for the same subject, I believe. Can anyone explain the differences between the two prizes?
Ответить
Nope🎉
Ответить
But what can you use the created compound for? And is this the best way to make it?
Ответить
Hearing the words column chromatography gives me PTSD from my silica layer cracking 5 TIMES IN A ROW. But as a Chem major I love watching these videos
Ответить
I wish I could understand the significance of this but... not there yet :) Cool video though, I presume :)
Ответить
One of the recipients was my Ochem professor. He was so excited about this, even before winning. The party we threw for him at UCPH was amazing. Great professor
Ответить
how about the tetrazole producing ugi 4CR?
Ответить
I think on a technicality this is a “copper-catalysed azine-alkyne coupling” rather than a click?
Ответить
Advice for column chromatography:
Layer number 1: cotton wool
Layer number 2: sea sand for flat surface
Layer number 3: slurry of silica gel
Layer number 4: pack the silica gel
Layer number 5: sea sand (flat surface is required for good separation)
Layer number 6: add the dilluted crude product with glass pasteur pipette (easy to clean) and use circular motion with your hands to evenly distribute the diluted crude product (gently add the diluted crude product to do not mess up the evenly distributed sea sand surface)
Layer number 6 alternative: you can use celite as the carrier (flat surface is required for good separation)
Layer number 7: another sea sand layer (flat surface is required for good separation)
Gently add the eluent mixture to the column and have fun.

The number of steps involved in this is dizzying. Where they all Really necessary to obtain this product?
Given the length of time involved, and the number of chemical processes to obtain this final product, I'm missing the advantage that "click" chemistry is supposed to provide.

in circumstances where your crude material does not completely dissolve in your eluant, it often helps to load your material onto celite or silica, by taking a solution of your crude (in e.g. acetone), forming a suspension with the celite powder, and then removing all solvent under vacuum (with or without heating). You can then add the resulting free-flowing powdered solid directly atop the bed of sand at the top of your column
Ответить
This looks like an easier way for adding a triazole ring let's say... a benzodiazepine ring ? Making it the triazolo version of the bzd... i have no diploma in organic chemistry (only process engineering so no real organic chem) so please correct me if i'm wrong.
To me it looks like a really easy way for pharma companies To produce more potent, more imparing, shorter lasting triazolobenzo compound (which are more addictive and worse for many other reasons) than the current synthesis available (involving hydrazine I guess)
After the whole thing is done, how easy it is To remove the tosyl part of the molécule from the triazolo-R group part ?
I mean yay research leads to overall better production methods, can't argue with that. But who leads research in this field? If it is, a way of making on a huge industrial scale, addictive drugs like xanax or ativan (or worse) ; i dont think it deserves a nobel prize.
If a benzodiazepine ring cannot become a triazolobenzo from this click chemistry for reasons i ignore (more selectivity for other sites of the molecule or very hard To make precursor, Idk i'm an amateur) then it deserves a nobel prize, since it helps science going forward.
But if it is an amazing discovery lead by S@ /|/ |} ○Z or B@y3r To increase their income from making and distributing lifewreking drugs ; then my initial "yay" is less enthousiastic.
I still think the chemistry behind it is awesome and i'm glad when people develop better technique in org chem. I'm just afraid of who paid for this research and would be disappointed if it is for drug manufacturing optimisation rather than making new molecules more available (for curing cancer and all).
Just adding my grain of ethic though. Wonder what people more advanced in those subjects (pharmacology & org synth) think about this and i would love To have other points of views
I apologize in advance for the bad english, it isn't my first language.
Great vidéo and good explainations, congrats :)

If this is the "relatively simple" method i can only imagine how insane the normal method is. Probably requires several blood oaths to satan and extraction using unicorn phlegm
Ответить
The idea is worth the Nobel price but the way it "clicks" is far inferior to biochemical bond breaking/formation.
Ответить
The workup and purifications required for any sort of synthetic chemistry are why I'm a computational chemist.
Ответить
chemistry of dipole particleish things
\

What is low radiation EMT magnetic oil?
Ответить
no... but you did a good 42 brother
Ответить
I use click chemistry to couple PEG to MOF drug carriers, where the azide of a organic modulator bound to the MOF reacts with a terminal alkyne on PEG. Really cool and easy stuff that you can monitor with FT-IR
Ответить
Love your channel, keep up the great work 👍🏼 👏
Ответить
Well done.
Ответить
Simple way🙂
Ответить
Dude you are amazing
Ответить
I use this technique in my lab all the time. We've engineering a protein with a non canonical amino acid with an azide allowing it to be coupled to an alkyne linker
Ответить
what kind of vacuum pump do you have? I mean the model
Ответить
Hey, but we still need so many solvents, etc. I think then it's not a very green reaction? Or there was a reaction before for this product which was not green at all even worse than this one
Ответить
Do you do this in a home lab?
Ответить
Man that was fun
Ответить
Absolutely remarkable, Nobel prize well deserved on this one
Ответить
Why dont you use cu+1 directly, ?
Ответить
As easy as a click they say.
Ответить
I was hoping you would just mix two things and get the new product. I think there is another Nobel prize waiting for someone who would actually make this process much simpler.
Ответить
Very good video. I did my graduate work on organic azide chemistry. I liked you video presentation very much.
Ответить
This is a really cool reaction, and I appreciate all the detail you took creating this video. I wonder how easy it is to release the tosyl group to add other substituents to the triazole ring. Also, this looks like there is a bit of Diels-Alder effect causing this "snapping into place" that's aiding the facility of this reaction. Pure chemistry doesn't get a lot of love these days from the Nobel committee.
Ответить
Thanks
Ответить
Did I miss the explanation of the mechanism?
Ответить
Круто
Ответить
Hype
Ответить
dawg what just happened
Ответить
This is one of the best videos of practical organic chemistry I probably ever saw. Click chemistry is cool.
Ответить